Intact Fish Skin for Operating Room Success
Managing surgical and trauma wounds
Our intact fish skin grafts for Operating Room use have helped thousands of healthcare providers with some of their most difficult cases.
Kerecis SurgiClose® and SurgiBind® Products
SurgiClose is an intact fish skin graft used for supporting tissue regeneration on surgical, traumatic and acute wounds.
SurgiBind is Intact fish-skin intended for surgical implantation. The product can be used to reinforce soft tissue where weakness exists.
The Kerecis fish skin graft are supplied in multiple shapes, variants and sizes and contain natural skin elements and structure.
The product classifies as a medical device and consist of a full thickness fish-skin that has been processed using Kerecis’ proprietary EnviroIntact™ method. The product is not subject to the May 31, 2021, FDA 351 regulation.

SurgiClose Micro
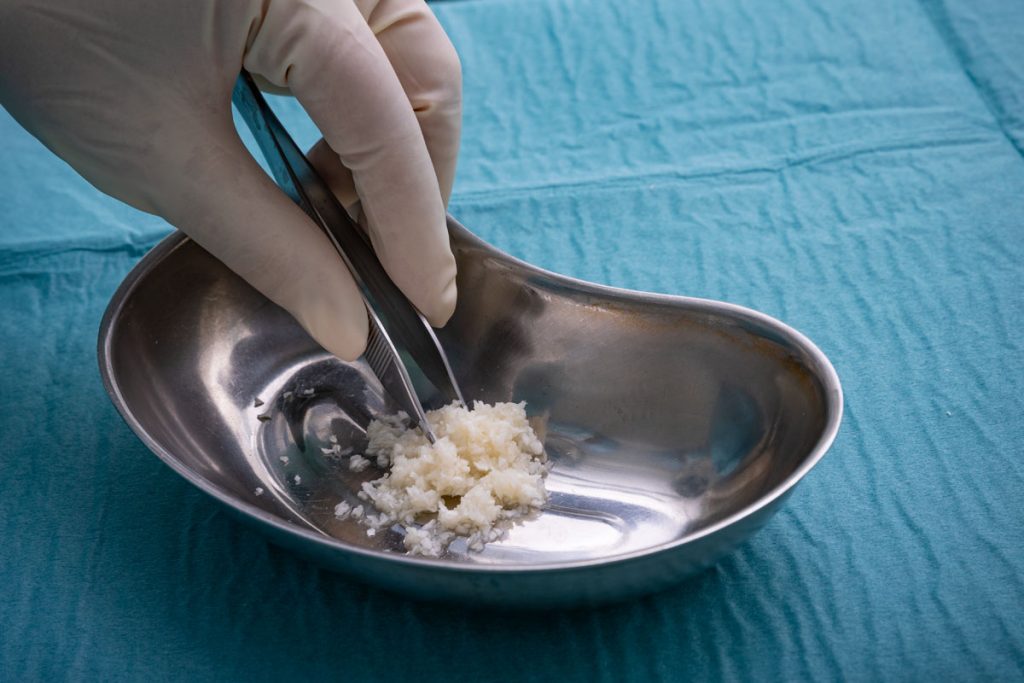
Intact fish skin graft is cut into smaller sheets using a process that preserves the integrity and composition of the material.
SurgiBind Fenestrated
Intact fish skin graft fenestrated to allow for easier flow through of liquids
SurgiClose Meshed 2:1
2:1 pre-meshed intact fish skin graft designed to expand and cover larger wounds. Provides a viable clinical and economic solution for large wounds
Contact Your Dedicated Sales Professional
Our Vision
To extend life by supporting the body’s own ability to regenerate.
![]()
I’ve used Kerecis on Donor Sites, and it’s the first time my patients haven’t complained about pain.
– Dr. Daniel Lozano MD
Benefits of Using SurgiClose & SurgiBind
Experience the Benefits of SurgiClose and SurgiBind in the Operating Room
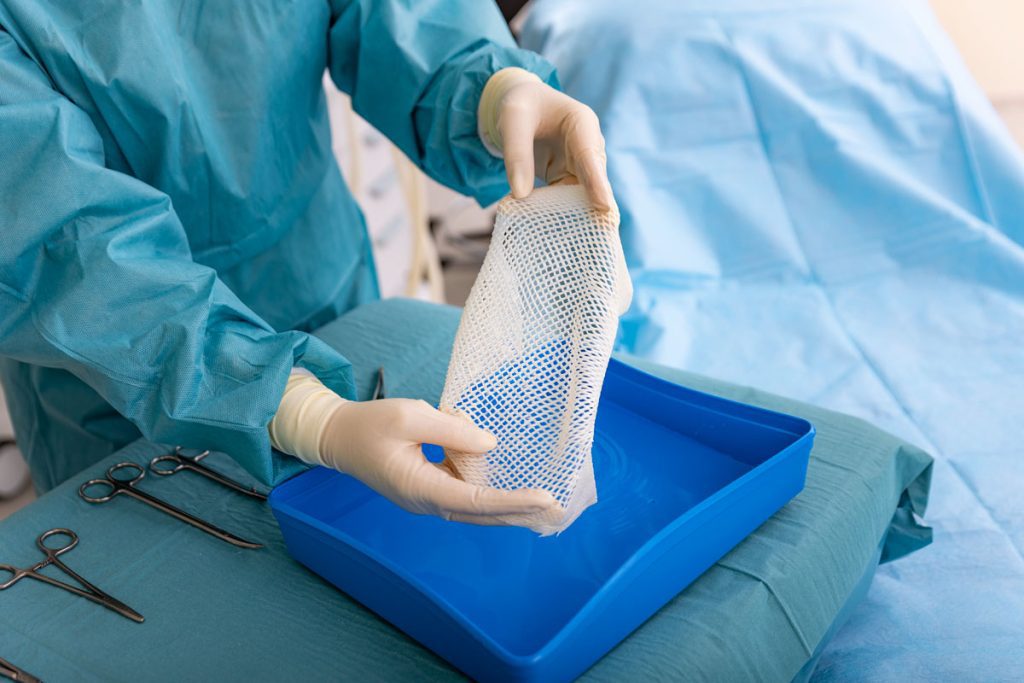
Reduced Procedure Time – All Kerecis products have a 3-year shelf life and do not need to be tissue tracked or refrigerated. When applying, hydrate our product in a sterile saline for 30 seconds and simply cut or stretch product to the size of the wound and fixate with your chosen method.
Enhanced Wound Healing – Compared to other skin substitutes. We are seeing faster healing times compared to dHACM. Check out our studies to learn more.
Versatile Applications – Kerecis can be utilized in a variety of specialties with head-to-toe indications. Kerecis offers a variety of sizes and variations to fit your needs.
Built on Sustainability – Kerecis’ manufacturing takes place in the Westfjords of Iceland, an area known for its commitment to environmental protection. We create life-saving value by transforming by-products from the fishing industry into medical devices. Our products are made from locally procured, certified, sustainably sourced North Atlantic Cod. Our manufacturing requires no harsh chemicals that might harm the environment, and it uses exclusively clean, renewable energy.
See how Kerecis saved this patient's life with Necrotizing Fasciitis
![]()
What I do with Hidradenitis Suppurativa is completely close with Kerecis.
– Dr. Michael Romberg MD
Need more information?
From the town of Isafjordur in northwest Iceland, Kerecis develops, manufactures, and sells patented fish skin soft tissue regeneration products that have regulatory approval in the United States, Europe and several other jurisdictions.
Other Kerecis Products

GraftGuide™
Mano
The intact fish-skin graft in the shape of the hand to easily allow application on its complex 3D structure. Available in two sizes, medium and large, and in either left or right versions, for use in palmar or dorsum applications. Designed to reduce operation time when managing hand burn injuries.
GraftGuide™
Micro
The intact fish-skin graft is cut into smaller sheets using a process that preserves the integrity and composition of the material
GraftGuide™
Meshed 2:1
2:1 pre-meshed intact fish-skin graft designed to expand and cover larger wounds. Provides a viable clinical and economic solution for large wounds.
User Information
Indication for SurgiClose
- Trauma Wounds
- Abrasions
- Lacerations
- Skin Tears
- Surgical Wounds
- Donor site/grafts
- Post-Mohs surgery
- Post-laser surgery
- Podiatric
- Dehiscence
Indication for SurgiBind
- For implantation to reinforce soft tissue where weakness exists, in patients requiring soft tissue repair, or reinforcement in plastic or reconstructive surgery.
The product derives from a from a fish source and should not be used in patients with a known allergy to fish or sensitivity. Note that fish-allergy is not the same as shellfish-allergy.
The product and its variations are not available in all countries and may be known under different trade names. Products, product names, indications and claims vary between jurisdictions. Contact your Kerecis representative if you have questions about the availability of Kerecis products in your area.
Always make sure to check with your local health care professional and read the actual printed package information prior to use. Information on this website might not be current and might not be an accurate reflection of current product labeling.
This website is not intended to provide diagnosis, treatment or medical advice. Products, services, information and other content provided on this website, including information that may be provided on directly or by linking to third-party websites, are provided for informational purposes only. Information on this website should not be considered as a substitute for advice from a healthcare professional.
Consult with a physician or other healthcare professional regarding any medical or health related diagnosis or treatment options.
A surgeon must always rely on his or her own professional clinical judgment when deciding whether to use a particular product when treating a particular patient. Kerecis does not dispense medical advice and recommends that surgeons be trained in the use of any particular product before using it in surgery.
The information presented on this website are intended to demonstrate the Kerecis product offerings. A surgeon must always refer to the package insert, product label and/or instructions for use before using any Kerecis product.
