SurgiBind is intact fish skin that is used for soft tissue reinforcement and grafting. It is based on the Kerecis graft technology that is FDA cleared and one of the leading products in the market for tissue regeneration and treatment of chronic, trauma and burn wounds.
SurgiBind provides an optimal structure for cell and vascular ingrowth. Fish skin graft has been shown to facilitate 20x more cell ingrowth than amniotic tissue and to promote revascularization and remodeling faster than a mammalian-sourced tissue. [1] [3]
SurgiBind® Benefits
Rapid incorporation [3]
Fast cell ingrowth [1]
Accelerated neovascularization[3]
Faster wound closure[5]
Natural bacterial barrier [1]
SurgiBind Product Variations
Tendon Protect
SurgiBind Tendon Protect is a solid sheet of intact fish skin intended for the protection and management of newly repaired tendons.
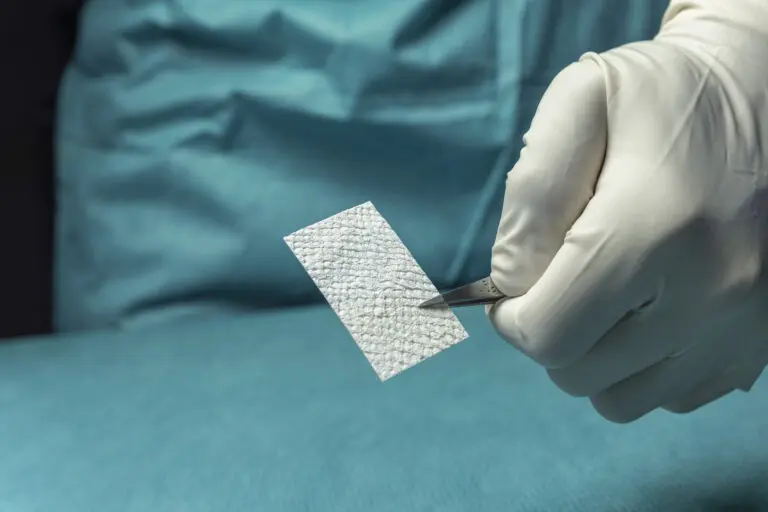
Solid
SurgiBind is a solid sheet of intact fish skin that is indicated for implantation and can be used to reinforce soft tissue where weakness exists, in patients requiring soft tissue repair, or reinforcement in plastic or reconstructive surgery.

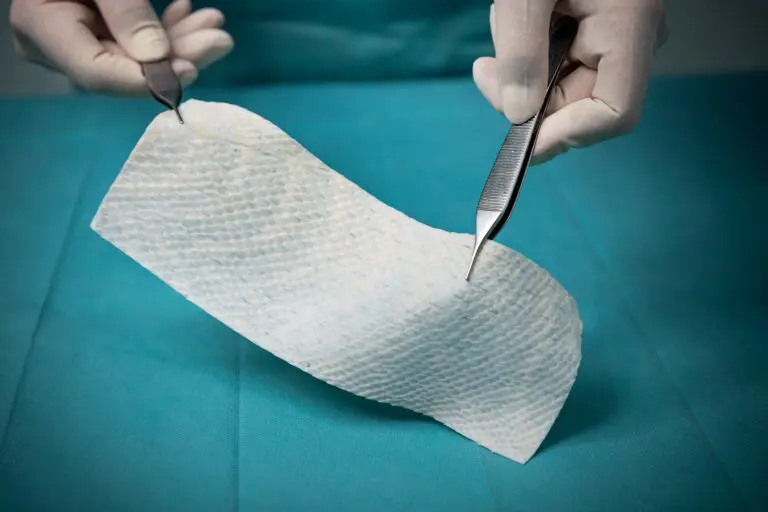
Fenestrated
Pre-fenestrated intact fish skin graft for supporting healing and tissue regeneration of chronic wounds.
Indications
For implantation to reinforce soft tissue where weakness exists, in patients requiring soft tissue repair, or reinforcement in plastic or reconstructive surgery.
SurgiBind Tendon Protect is indicated for the management and protection of tendon injuries in which there has been no substantial loss of tendon tissue.
Need more information?
From the town of Ísafjörður in northwest Iceland, Kerecis develops, manufactures, and distributes patented fish-skin medical devices that support soft tissue regeneration in the body, with regulatory clearance in the United States, Europe, and beyond.
Important Information
Magnusson, et al. Regenerative and Antibacterial Properties of Acellular Fish Skin Grafts and Human Amnion/Chorion Membrane: Implications for Tissue Preservation in Combat Casualty Care. Mil. Med. 182, 383–388 (2017).
Magnusson, S. et al. Decellularized fish skin: characteristics that support tissue repair. Laeknabladid 101, 567–573 (2015).
Stone R 2nd, Saathoff EC, Larson DA, et al. Accelerated Wound Closure of Deep Partial Thickness Burns with Acellular Fish Skin Graft. Int J Mol Sci. 2021;22(4):1590.
Patel, M. & Lantis II, J. C. Fish skin acellular dermal matrix: potential in the treatment of chronic wounds. Chronic Wound Care Manag. Res. 6, 59–70 (2019).
Badois, N. et al., Acellular fish skin matrix on thinskin graft donor sites: a preliminary study. J Wound Care 28. 624–628 (2019).
Dardari D, Piaggesi A, Potier L, Sultan A, Diener H, Francois M, Dorweiler B, Bouillet B, M’Bemba J, Chaillous L, Clerici G, Kessler L, Wetzel-Roth W, Storck M, Davidsson OB, Baldursson B, Kjartansson H, Lantis JC, Charpentier G. Intact Fish Skin Graft to Treat Deep Diabetic Foot Ulcers. NEJM Evid. 2024 Oct 4, doi: 10.1056
