As a plastic and reconstructive surgeon, you take on some of the most complex surgical procedures. You need a tissue-repair product that helps you navigate the reconstructive ladder with confidence to support patient outcomes in challenging cases.
Kerecis® intact fish-skin grafts provide the robust three-dimensional structure and natural molecular content needed to rapidly facilitate cellular ingrowth, maintain a bacterial barrier, and promote tissue regeneration.
Because Kerecis is minimally processed and retains the fish’s natural skin structure – which is remarkably similar to human skin – it integrates quickly, enabling you to reduce time to definitive reconstruction or unlock options lower on the reconstructive ladder.
Kerecis for Plastic and Reconstructive Surgery
Reconstruct over exposed structures [1]
Promote faster healing in compromised tissue beds [2,3]
Limit secondary contracture to optimize functional and cosmetic outcomes [4-6]
Reconstruct Over Exposed Structures, Including Bone & Tendon
Promoting granulation on surgical sites with exposed bone and tendon is a key challenge for plastic surgeons. Kerecis is indicated for use on full-thickness wounds with exposed bone and tendon and is clinically proven to fast-track healing on these tough sites, equipping surgeons to potentially reduce from flap to graft.
A 2024 multi-center RCT found that Kerecis products promoted faster healing for severe wounds with exposed bone and tendon compared to standard of care. [1]
Promote Faster Healing in Compromised Tissue Beds
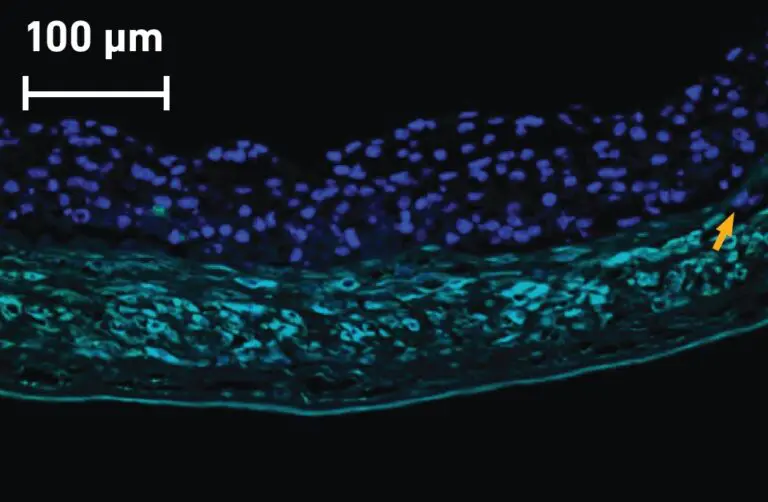
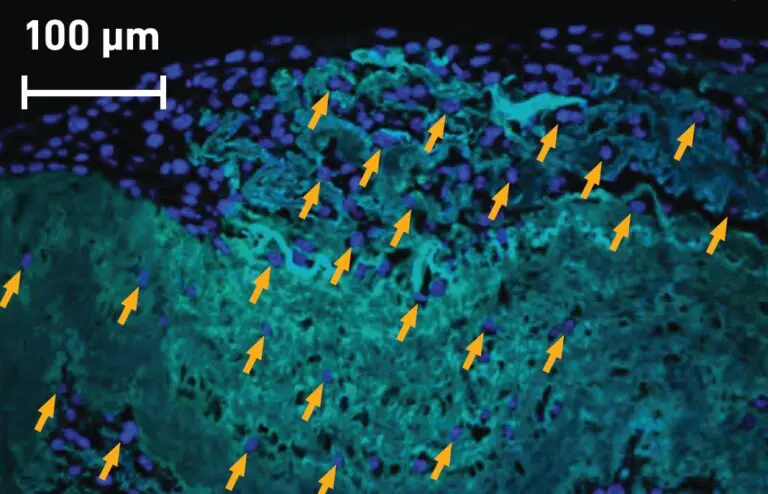
Our fish-skin grafts’ intact ECM components help enhance healing in damaged or hypovascularized tissue beds by rapidly incorporating and facilitating cellular ingrowth,[2,3] creating a healthy area ready for closure by secondary intention, autograft, or flap.
Kerecis Fish-Skin Graft has Significantly More Cell Ingrowth than Amnion / Chorion Membrane
Limit Secondary Contracture to Optimize Functional & Cosmetic Outcomes
Poor graft integration and disorganized tissue remodeling can compromise patient outcomes. Kerecis’ 3D tissue structure provides a robust platform for fibroblast infiltration, promoting fast integration. This may help limit scarring, adhesion, and secondary contracture, [4,5,6] which can help restore and maintain function such as range of motion.
Surgical sites treated with Kerecis have also demonstrated improved skin pliability, pigmentation, vascularity, and hydration levels.[6] A 2021 feasibility trial found that full-thickness burns treated with Kerecis had similar functional and cosmetic outcomes as burns treated with cadaver skin one year after treatment.[6]
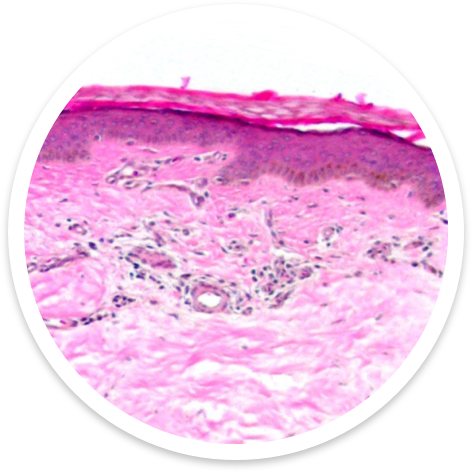
Navigate the Reconstructive Ladder with Confidence
See examples of how Kerecis can support you below.
Plastic and Reconstructive Surgery Case Studies
Bridging to Skin Graft
Patient Presentation
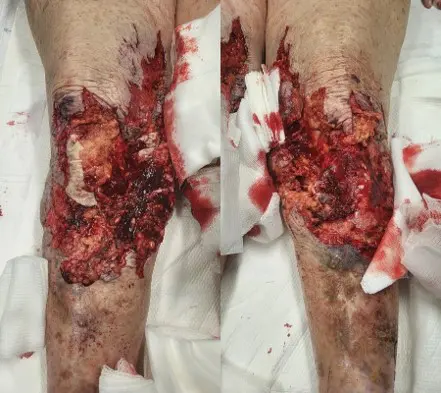
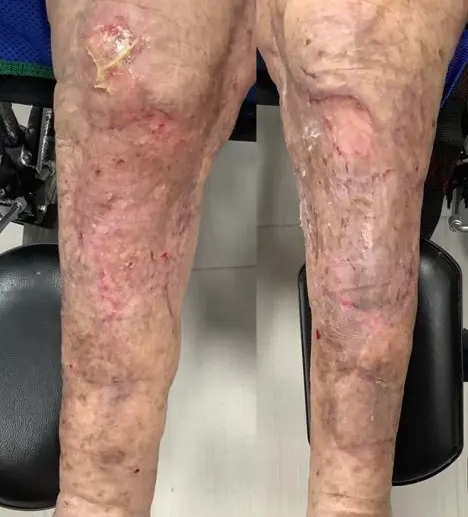
88-year-old female presented to ED as trauma after a motor vehicle crash. Sustained extensive degloving injury to BLE.
Underwent washout of LE wounds with placement of negative pressure wound therapy (NPWT) to both legs by general surgery on three occasions.
Surgical Intervention & Kerecis Application
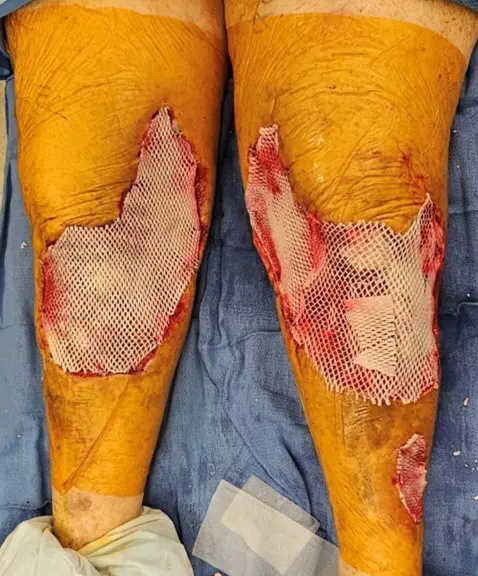
First Application: SurgiClose Micro and Meshed 2:1, done by plastic surgery
Second Application: SurgiClose Meshed 2:1, done by plastic surgery
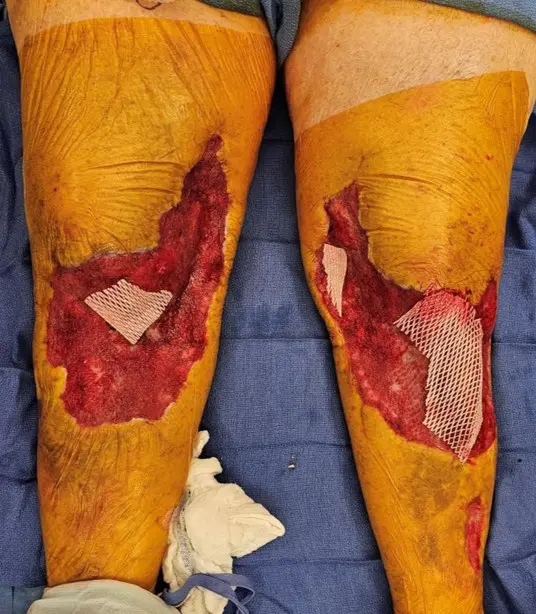
Excellent take of Kerecis with nearly entirely covered patella tendon, muscle fascia, and tibia periosteum. Very minimal slough. Significant reduction in pain with NPWT dressing changes.
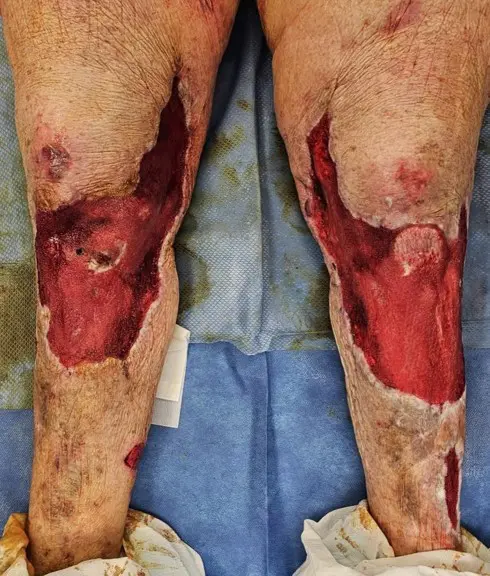
Clinical Outcome
Patient had complete closure of entire wound, including areas of exposed patellar tendon, tibial periosteum, and exposed muscle fascia. Dramatic improvement in pain after Kerecis placement with decreased pain medication usage.
Healing by Secondary Intention
Patient Presentation
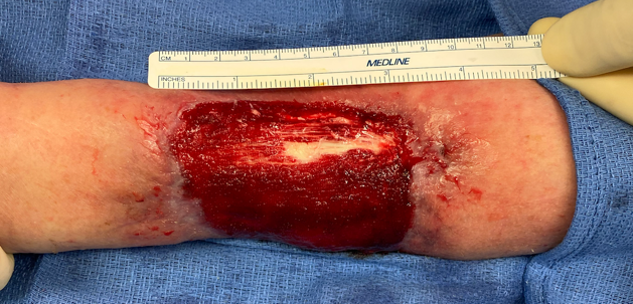
61-year-old female presented to the clinic on February 11 after multiple dog bites to left upper extremity, requiring multiple debridements – ultimately, resulting in a dorsal forearm wound and exposed extensor tendons. Reconstructive options with other skin substitutes had failed.
Surgical Intervention & Kerecis Application
SurgiClose was used over the exposed extensor tendon on the dorsal forearm without overlying peritenon. Kerecis was not applied to the remainder of the wound bed as it had healthy granulation tissue.
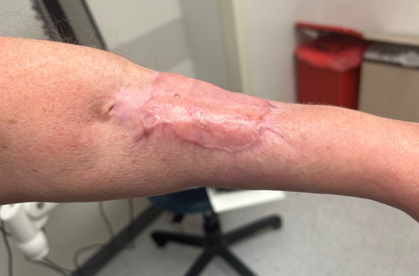
Clinical Outcome
The patient’s wound healed with no pain and minimal loss of wrist or finger range of motion. Patient was highly satisfied.
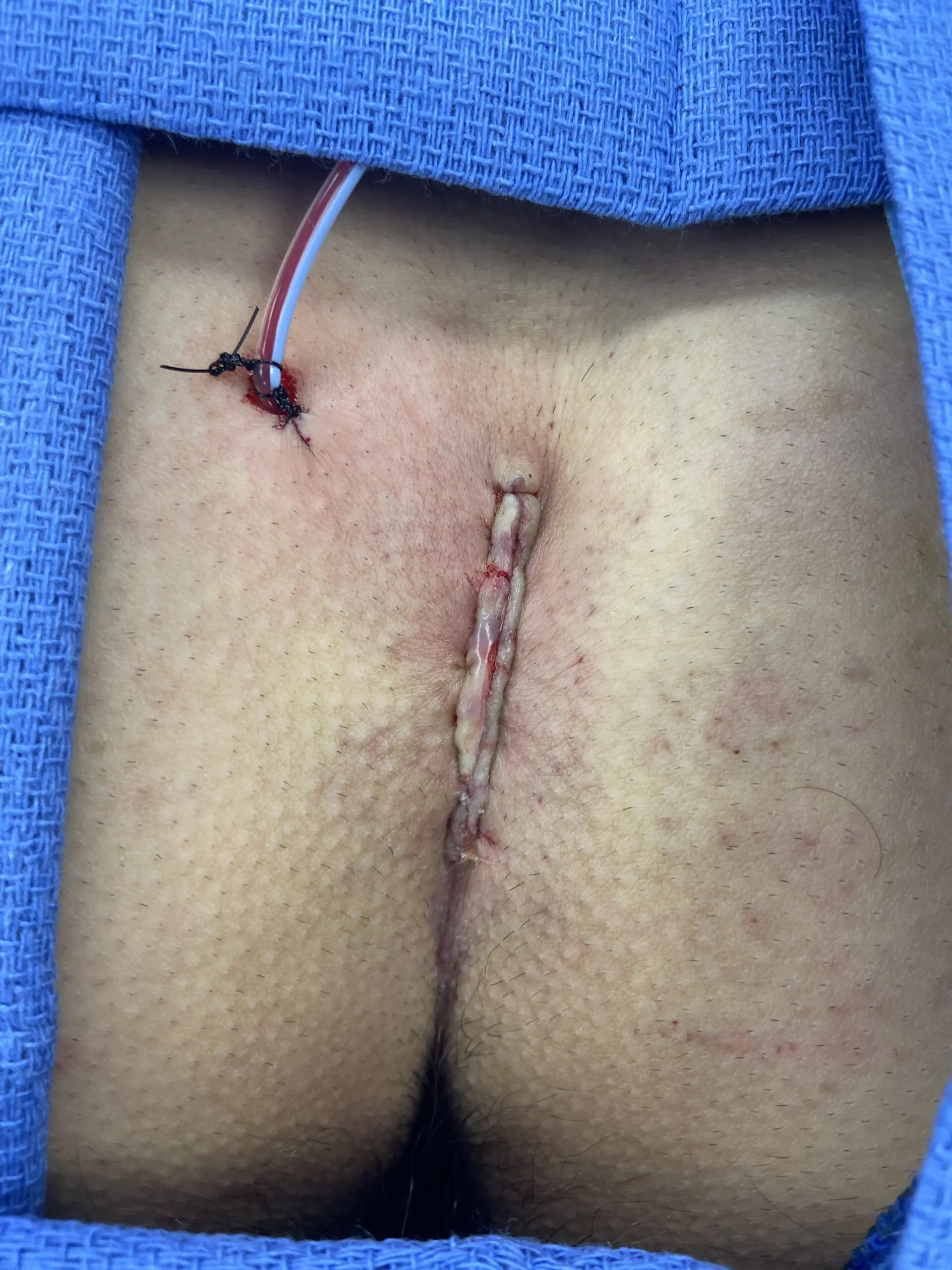
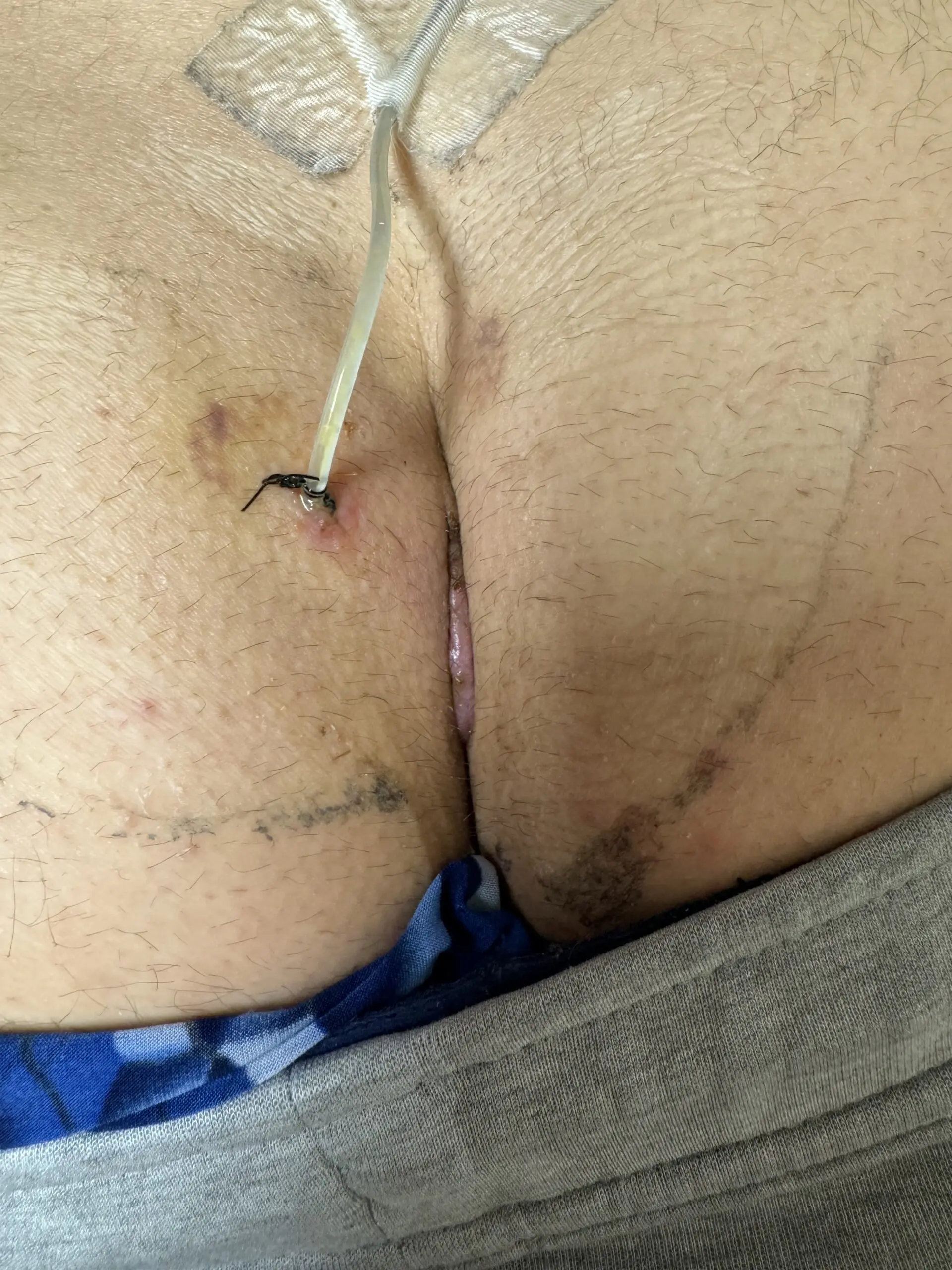
Primary Closure
Kerecis can be applied under the suture line to support reinforcement and reduce the risk of dehiscence. In the case series below, five patients undergoing pilonidal cyst excision fully healed with Kerecis placed as an underlay to the skin closure.
Patient Presentation
5 patients undergoing pilonidal cyst excision were included in this clinical series
All patients were male and the average age was 26 (range 22-29).
All patients received 2 grams of cephazolin preoperativel
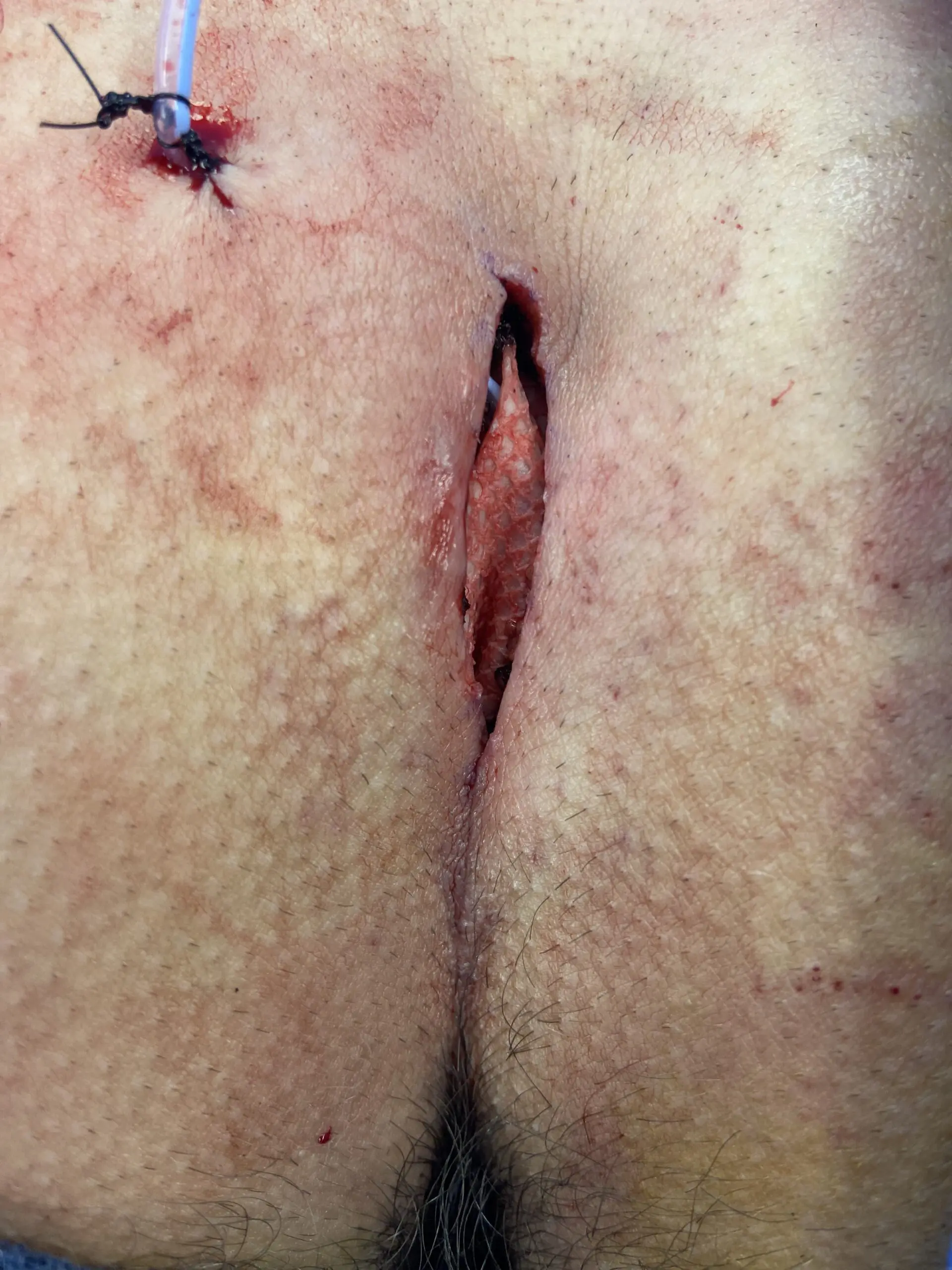
Surgical Intervention and Kerecis Graft Application
SurgiBind reconstituted in a vancomycin/saline solution was placed as an underlay to the skin closure.
Clinical Outcome
All wounds healed uneventfully, and all sutures were removed at Day 10.
No wounds demonstrated any signs of infection or wound dehiscence.
This small pilot study suggests the possible role of full thickness fish skin underlay in preventing wound dehiscence in patients undergoing primary closure after pilonidal cyst excision
Discover the Benefits
Important Information
Dardari, et.al. Intact Fish Skin Graft to Treat Deep Diabetic Foot Ulcers. The Odinn Trial. NEJM Evidence, 2024: 5. DOI: 10.1056/EVIDoa2400171.
Yoon J, Dogeon Yoon, Lee H, et al. Wound healing ability of acellular fish skin and bovine collagen grafts for split-thickness donor sites in burn patients: Characterization of acellular grafts and clinical application. Int J Biol Macromol. 2022;205:452-461. doi:10.1016/j.ijbiomac.2022.02.055 5.
Kotronoulas A, Jónasdóttir HS, Sigurðardóttir RS, Halldórsson S, Haraldsson GG, Rolfsson Ó. Wound healing grafts: Omega-3 fatty acid lipid content differentiates the lipid profiles of acellular Atlantic cod skin from traditional dermal substitutes. J Tissue Eng Regen Med. 2020;14(3):441-451. doi:10.1002/ term.3005
Lacqua, M et al (2021). The use of intact fish-skin wrap in preventing adhesions during tendon repair: A report of two cases. Edorium J Orthop 2021;7:100018O03ML2021.
Wallner, C. et al. (2022). The use of intact fish skin as a novel treatment method for deep dermal burns following enzymatic debridement: A retrospective case-control study. European Burn Journal, 3(1), 43–55.
Shupp J, McLawhorn M, Moffatt L (2021). Fish Skin Compared to Cadaver Skin as a Temporary Coverage and Wound Bed Preparation for Full Thickness Burns: An Early Feasibility Trial. J Burn Care Res. 42:S124-S124. doi:doi:10.1093/jbcr/irab032.201
Need more information?
From the town of Ísafjörður in northwest Iceland, Kerecis develops, manufactures, and distributes patented fish-skin medical devices that support soft tissue regeneration in the body, with regulatory clearance in the United States, Europe, and beyond.